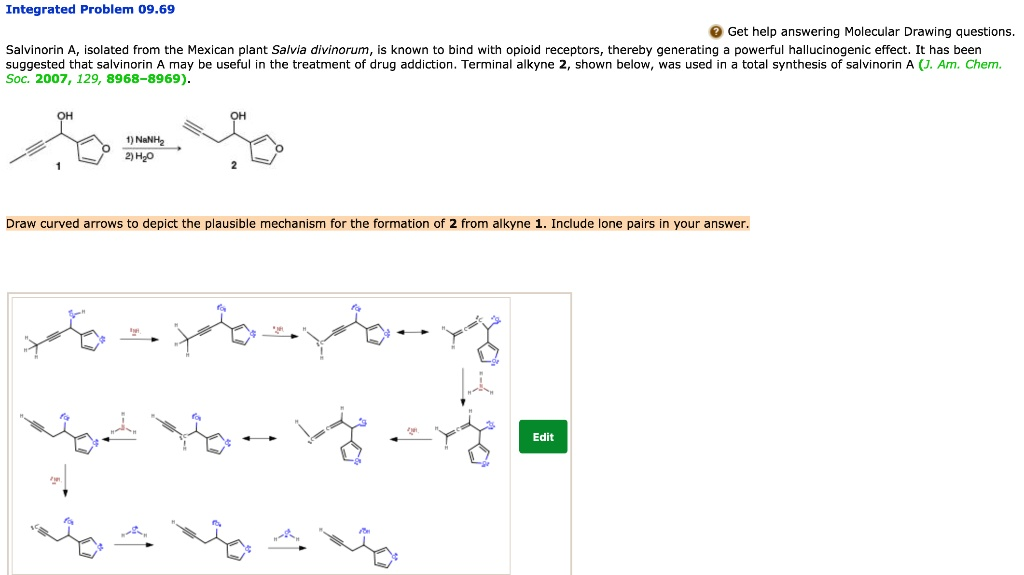
Integrated Problem 09.69 Get help answering Molecular Drawing questions. Salvinorin, isolated from the Mexican plant Salvia divinorum, is known to bind with opioid receptors, thereby generating a powerful hallucinogenic effect. It has been suggested that salvinorin may be useful in the treatment of drug addiction. Termina Ikyne, shown below, was used in the total synthesis of salvinorin (J Am Chem Soc, 2007, 129, 8968-8969). 1) NeNkz 2Hzo Draw curved arrows to depict the plausible mechanism for the formation of Ikyne from NeNkz. Include lone pairs in your answer.
The Correct Answer and Explanation is:
To explain the formation of the terminal alkyne (Ikyne) from its precursor (NeNkz), we are dealing with a classic deprotonation of a terminal alkyne followed by reprotonation in aqueous work-up. The reagents are sodium amide (NaNH₂) followed by water.
Step-by-Step Mechanism:
- Base Deprotonation: Sodium amide (NaNH₂) dissociates in solution to form Na⁺ and NH₂⁻. The lone pair on the nitrogen of the amide ion (NH₂⁻), a strong base, attacks the terminal hydrogen of the alkyne.
- Formation of Acetylide Ion: The terminal hydrogen, being slightly acidic due to the sp-hybridized carbon’s electronegativity, is removed. A lone pair remains on the terminal carbon, forming a carbanion. This results in a resonance-stabilized acetylide ion, which is nucleophilic and can participate in further transformations.
- Reprotonation (Work-up): Water (H₂O) is added to quench the reaction. The lone pair on the negatively charged carbon attacks a proton from water, restoring the terminal hydrogen and neutralizing the acetylide ion, completing the formation of the terminal alkyne Ikyne.
Explanation:
This sequence is a classic acid-base reaction followed by a quench. The acidity of the terminal alkyne’s proton is sufficient for abstraction by the strong base NH₂⁻. This step generates a stabilized acetylide anion. The negative charge on carbon is relatively stable due to the high s-character of the sp-hybrid orbital. In the final step, water donates a proton to the carbanion, forming the terminal alkyne product.
Every curved arrow should originate from a lone pair and point to either a proton (in the case of abstraction) or a hydrogen atom bonded to oxygen (in the case of quenching), maintaining proper electron flow and charge balance in the mechanism.
