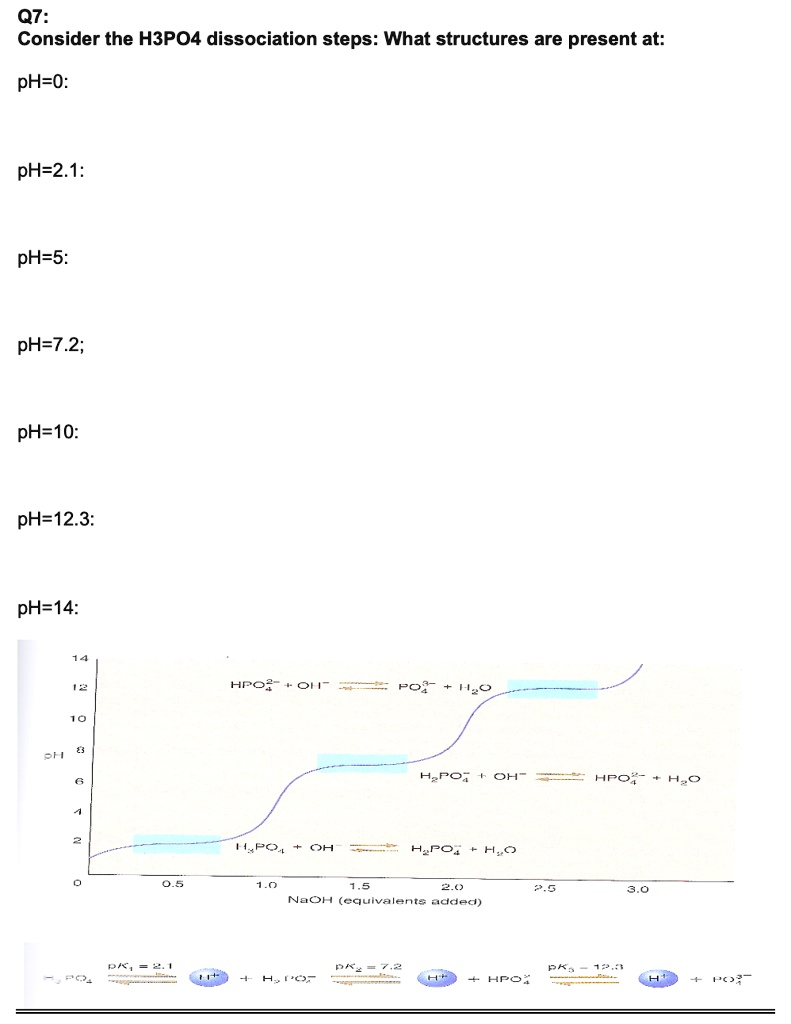
Consider the H3PO4 dissociation steps: What structures are present at: pH-0: pH-2.1: pH-5: pH-7.2; pH-10: pH-12.3: pH-14: H3PO4? OH- PO4- 1120 BH H3PO4; OH- H2O PO4 OH H3PO4; H2O 7-5 20 NaOH (equivalent added) 75 PA 774 70 A”‘ H3PO4; ATC
The Correct Answer and Explanation is:
Based on the provided titration curve and pKa values for phosphoric acid (H3PO4), the predominant chemical structures at each specified pH are as follows:
- pH=0: H3PO4 (phosphoric acid)
- pH=2.1: H3PO4 and H2PO4- (in equal concentrations)
- pH=5: H2PO4- (dihydrogen phosphate)
- pH=7.2: H2PO4- and HPO4^2- (in equal concentrations)
- pH=10: HPO4^2- (hydrogen phosphate)
- pH=12.3: HPO4^2- and PO4^3- (in equal concentrations)
- pH=14: PO4^3- (phosphate)
Explanation
Phosphoric acid (H3PO4) is a polyprotic acid, meaning it can donate more than one proton in a stepwise manner. The provided titration curve and pKa values illustrate these sequential dissociation steps. The predominant chemical species in a phosphate solution is determined by the solution’s pH relative to the three pKa values: pKa1 = 2.1, pKa2 = 7.2, and pKa3 = 12.3.
The pKa value represents the pH at which an acid and its conjugate base are present in equal concentrations. This principle, derived from the Henderson-Hasselbalch equation, is key to determining the structures at different pH levels.
At a very low pH, like pH 0, the environment is highly acidic. This pH is well below the first pKa of 2.1, so the equilibrium strongly favors the fully protonated form, H3PO4.
When the pH equals the first pKa, at pH 2.1, the solution is at the midpoint of the first buffering region. Here, the concentrations of the acid (H3PO4) and its first conjugate base (H2PO4-) are equal.
At pH 5, the pH is above pKa1 but below pKa2. This means the first proton has mostly dissociated, but the second has not. Therefore, the dihydrogen phosphate ion, H2PO4-, is the major species.
Similarly, when the pH equals the second pKa, at pH 7.2, the system is at the center of the second buffer zone. The concentrations of the second acid-base pair, H2PO4- and HPO4^2-, are equal. This buffer system is biologically significant.
At pH 10, the pH is well above pKa2 but still below pKa3. This indicates the second proton has dissociated, making the hydrogen phosphate ion, HPO4^2-, the predominant structure.
At pH 12.3, the pH equals the third pKa. Here, the final acid-base pair, HPO4^2- and PO4^3-, are present in equal amounts.
Finally, at a very high pH, such as pH 14, the environment is strongly basic. This pH is significantly above the last pKa, driving the equilibrium to the fully deprotonated form, the phosphate ion, PO4^3
