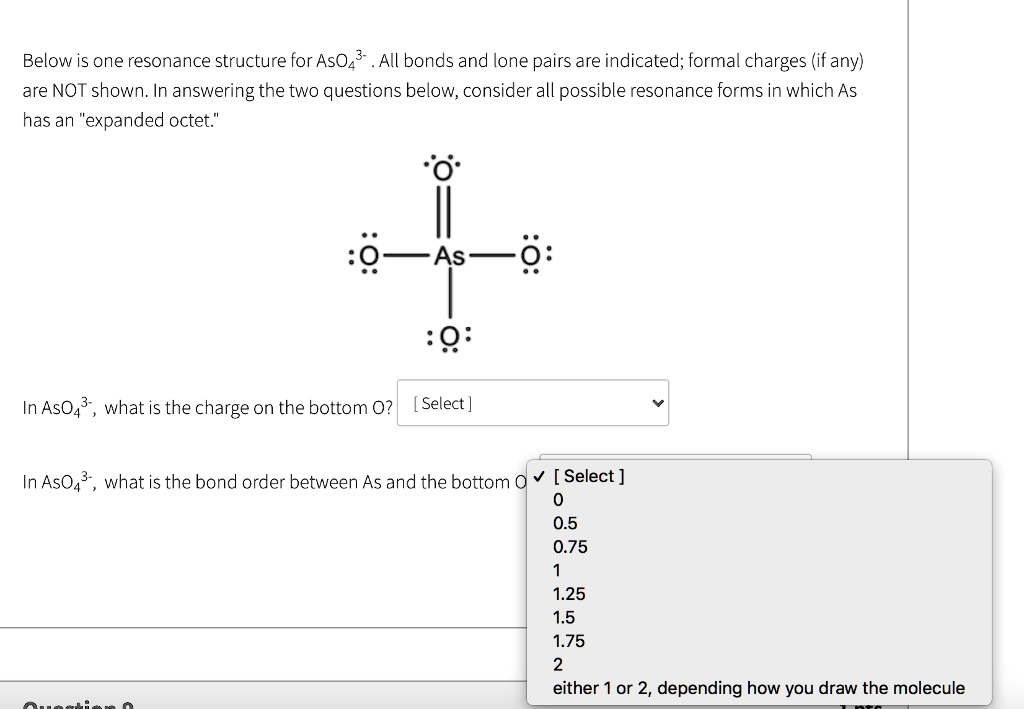
Below is one resonance structure for AsO
. All bonds and lone pairs are indicated; formal charges (if any) are NOT shown. In answering the two questions below, consider all possible resonance forms in which As has an \”expanded octet.\” :O || :O—As—O: || :O: In AsO
, what is the charge on the bottom O? [Select] In AsO
, what is the bond order between As and the bottom O? [Select] 0 0.5 0.75 1 1.25 1.5 1.75 2 either 1 or 2, depending how you draw the molecule
The Correct Answer and Explanation is:
For the arsenate ion (AsO₄³⁻), resonance plays a key role in understanding both formal charge and bond order. The structure you’ve provided shows arsenic at the center with one double bond and three single bonds to oxygen atoms, each oxygen having its lone pairs.
1. Charge on the bottom oxygen: To determine formal charge, use the formula: Formal charge = valence electrons − (non-bonding electrons + ½ bonding electrons) For oxygen (6 valence electrons):
- The bottom oxygen forms one single bond to arsenic (2 bonding electrons)
- It also has six non-bonding electrons (three lone pairs)
So, Formal charge = 6 − (6 + ½×2) = 6 − (6 + 1) = −1
Thus, the charge on the bottom oxygen is −1.
2. Bond order between As and the bottom oxygen: Arsenic can have an expanded octet, allowing more than eight electrons around it. In AsO₄³⁻, resonance occurs as the double bond shifts among the four oxygen atoms. Although only one structure shows a double bond between As and a specific oxygen, the true molecule is a resonance hybrid where each As–O bond has the same character.
There are four resonance structures, each with a double bond to a different oxygen and single bonds to the remaining three. So, for the bond to any one oxygen (such as the bottom one): Bond order = total bond character / number of resonance forms = (1×2 + 3×1)/4 = 5/4 = 1.25
Therefore, the bond order between As and the bottom oxygen is 1.25.
In summary:
- Charge on bottom O: −1
- Bond order between As and bottom O: 1.25
This highlights how delocalization stabilizes the ion and equalizes bond character across all As–O connections.
